Droplet size
Mean droplet size is actually a measure of the overall surface area of the fluid being sprayed. The smaller the droplet size the greater the surface area of the spray for any given volume of fluid. If one halves the mean droplet size of any given spray then the surface area of the spray doubles. If one quarters it then it quadruples and so on (for a detailed explanation of why this is so click here).
Why is droplet size important
The surface area of a spray is a reasonable approximation to its overall reactivity. Reactivity means its ability to produce a chemical reaction or the spray’s ability to absorb or dissipate heat. So in heat transfer and chemical reaction sprays the droplet size is one of the most important elements in determining how well the spray will perform.
Droplet size can also be important when considering the overall entrainment of a fluid within a gas flow. Smaller droplets will get swept along in a moving flow more quickly and if the spray is too fine this could overload mist eliminators. If operating in windy conditions then finer sprays might be blown off target so an understanding of droplet size is important.
What affects droplet size?
Pressure
A simple rule that holds for all nozzles is that the higher the fluid pressure, the smaller the droplet size. For any given hydraulic nozzle the relationship between pressure and mean droplet size can be expressed as:
Where D is the mean droplet size at pressure 1(P1) and pressure 2(P2).
This gives an approximate relationship for comparing droplet sizes for any given nozzle, but there is no easy mathematical relationship for any generic nozzle as droplet size depends greatly on the design of the nozzle.
Spray pattern type
It is fairly obvious that solid stream sprays do not really have droplets at all. Flat fan patterns may form sheets of liquid without much atomisation or may produce coarsely atomised sprays. Full cone nozzles will produce the next level of atomisation with hollow cone nozzles producing the smallest droplets.
Spray angle
Very simply, for any given flow rate, the wider the spray angle, the smaller the droplet size will be. It’s easy to understand why - larger angles sprays simply have more space to distribute the droplets and so there is less chance of recombination and a greater opportunity to atomise.
Nozzle type
The design of the spray nozzle will obviously affect spray pattern type (flat fan, hollow cone etc) and this will affect droplet size, as discussed above, but even staying within a pattern type there is variation on levels of atomisation. For example, the spiral design nozzle will produce a full cone pattern that, for a given pressure, flow rate and spray angle, will produce smaller droplets than an axial whirl nozzle. How different nozzle designs will affect droplet size can be complex and all the details can’t be covered here, but it should be borne in mind that changing nozzle design type can change the consistency of seemingly identical sprays.
Specific gravity of the fluid
The specific gravity of a fluid will affect the overall flow rate achieved at the nozzle and hence will affect droplet size. For a given pressure, the higher the specific gravity the lower the flow rate, hence the lower the mean droplet size. We have software that can calculate these effects so we would advise calling us. But the formula that is generally accepted is
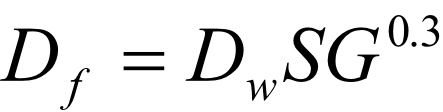
Df = the droplet size for the fluid in question
Dw = the droplet size for water for that particular nozzle
SG = specific gravity of the fluid
As specific gravity is often very close to 1 and the exponent is 0.3, the effect of this is generally very small.
Viscosity and surface tension
Fluids with higher viscosities than water will have higher mean droplet sizes for any given flow rate and pressure. Similarly fluids with higher surface tensions will form larger droplets. The interplay between the mechanical properties of fluids can get complex but software exists to perform droplet size calculations if the basic properties are fed in. We would advise speaking to one of our experts in order to obtain estimates for the effects of viscosity and surface tension on droplet size. But again the generally accepted formula is.
Where Df = modified droplet size for the fluid in question
Dw = Droplet size calculated for water
Vf = the viscosity of the fluid (viscosity in centipoise; water = 1.0 cP)
At first it would seem that the exponent of 0.2 would indicate that viscosity has a lower effect than specific gravity but it needs to be remembered that fluids can have viscosities 1000 times that of water and still be fluids where as even a super heavy fluid like mercury only has an SG of 13.
A similar equation and relationship exists for surface tension
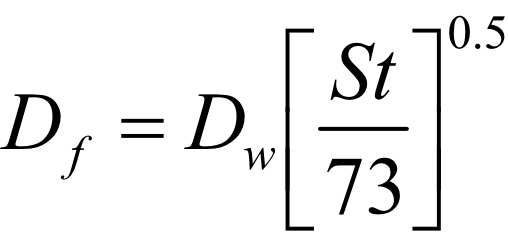
Where St is the surface tension of the fluid in Dynes /cm with water having a surface tension of 73 Dynes/cm at 20 degree C.
Please note that some text books disagree on the exponent for both the viscosity and surface tension equation.