The spraying of viscous fluids can represent some challenges. In the food industry there are many viscous fluids that need to be sprayed so clearly this a key concern in this sector.
Viscosity measures
Viscosity is a measure of a fluid's resistance to shear stress. It is an approximate measure of the 'thickness' of a liquid. Viscosity is measured in pascal seconds with water at 20 degrees C having a viscosity of 1.002 mPa.s. This is most commonly redefined as the unit Centipoise (cps) with 1 cps being equivalent to 1 millipascal second, i.e. water has a Cp of about 1 at room temperature.
Some common fluids and their cps
Water 1
Milk 3
Olive oil 40
Single cream 50
Double cream 120
Honey 1,800 – 3,000
Sour cream 15,000
Corn syrup 15,000
Effects of viscosity on fluid properties
As the viscosity of a fluid increases, it will tend to form larger droplets when sprayed. For very viscous fluids this will have a detrimental effect on coating applications as small droplet sizes help to ensure an even coating. It may also affect how well the coating adheres to the target. This is because sprays consisting of smaller droplets have a much larger surface area per volume than those made up of larger droplets. As such, finer sprays are better able to spread out on their target surface.
This effect is relatively small for fluids with viscosities below 10cps but becomes more pronounced with higher cps. For example, double cream (Cps 120) sprayed would form droplets about 2.6 times the size of that of water sprayed from the same nozzle. This would mean the spray would have 2.6 times lower surface area to volume ratio making it less likely to adhere to a surface and form an even layer.
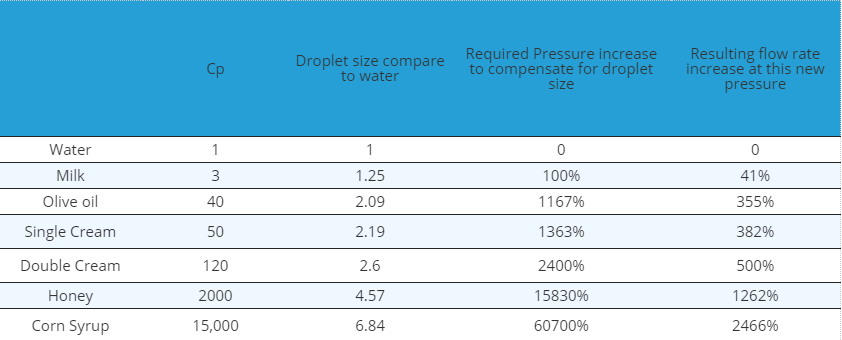
It may be possible to compensate for this by increasing the fluid pressure as increased pressure will reduce the droplet size, but spraying at higher pressures also increases the flow rates. The table below shows the droplet size increase compared to water and the corresponding required increase in fluid pressure to compensate, i.e. to achieve the same droplet size as one would expect when spraying water. The final column shows the resulting increased flow rate.
Viscous fluids will tend to form larger droplet sizes. A nozzle spraying water that will form a droplet size of Dw will form a droplet size of Df according to the formula below:
Droplet pressure formula
D1 – droplet size at P1
D2 – droplet size at P2
P1 – Pressure 1
P2 – Pressure 2
Droplet viscosity formula
Vf being the viscosity of the fluid in Cp as water has a Cp of 1
Df droplet size for fluid in question
Dw droplet size expected for water
Flow rate pressure formula
Q flow at pressure P
K – flow at 1 bar pressure in l/min
P – pressure in bar
Note: the exponent of 0.5 for P may vary depending on the nozzle used
Using the above formula we can produce this table for some common food sprays :
As can be seen from the table, droplet sizes increase dramatically with viscosity and there is not really any practical way to compensate for this as any pressure increase will have a knock on effect of increasing flow rate. In most applications other methods of atomisation for viscous fluids are required so air atomisers are used to achieve good droplet sizes but without the corresponding increases in pressures and flow rates.
Air atomising nozzles
Many air atomising nozzles mix the fluid and air inside the nozzle before ejection from the orifice. This is perfectly acceptable for low viscosity fluids like water, but with fluids much above 100cps problems occur. The larger droplet sizes and bubbles that form in the chamber severely impede the spray formation. This can lead to blockages and a severely degraded spray pattern. To overcome this problem external mix air atomisers should be deployed. These mix the air with the fluid stream after exit from the nozzle and so will not encounter the problems faced by internal mix atomisers.
Heat
The viscosity of a fluid will tend to lessen as it is heated. Conversely if certain liquids cool they will become more viscous. A practical example of this is chocolate. At room temperature it is solid and so clearly cannot be sprayed, but when heated it will become a liquid. At a little above melting point chocolate is still too viscous for practical spraying but raise its temperature a bit more and spraying becomes practical. A few degrees in temperature difference can mean the difference between a 1000cps fluid (not really practical for spraying) and a 100cps fluid which is able to be sprayed.
As increases in viscosity can be very rapid for even small temperature differences care must be given to such spraying systems. Undesired cooling of only a few degrees prior to spraying can impair the spraying process. As such heated water jackets are often used to maintain a stable fluid temperature right up to the nozzle orifice.
Viscosity vs stickiness
Although many viscous fluids are very sticky, technically viscosity is not the same as stickiness. Some very viscous fluids like sour cream are not that sticky while others, like honey, are very sticky. Stickiness may present other spraying challenges over and above those caused by viscous fluid.